Three reason to use Datatrace Pro
Powerful Reporting Capabilities
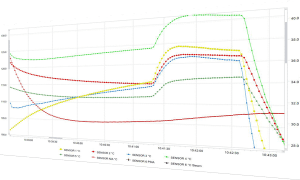
In the food processing industry, we need lethality data to know whether the product is safe to eat. Autoclave customers processing for sterilization will need reporting capabilities to display pass or fail criteria for their process. All of this information can now be graphed and displayed all on one report with just one extra click for a quick, easy-to-read review of the process for anyone to check the results. The robust reporting capabilities of the DT Pro software provide beautiful reports with graphs and calculations such as lethality, pre- and post-calibration verification displays of loggers before and after the process, and a placement view of the data loggers. This is an efficient solution for a reporting engine with no post-processing required. Time and energy spent post-processing data in excel or other calculation software can be put elsewhere while the DT Pro software does that work for you.
In Software Pre- and Post- Calibration Verification
There is a requirement to perform As Found results before use in EtO-sterilization, because once the gasses have affected the sensor, you need to prove that it was in calibration the next time it is used, right before use. Being able to do pre- and post-calibration on your own saves a lot of time and hassle. The software offers a “set it and forget it” type of calibration with industry standard controls and the ability to assess to a certain specified tolerance. It can calibrate loggers directly in the software and communicate with industry standard or leading metrology equipment. This saves time and energy while giving you the control over the calibration to determine which data points are being checked and verified. Your loggers will be kept within tolerance while removing hassle from the equation. Please note, it is still recommended that you receive factory calibration once per year, but performing it in house simplifies the process.
21CFR Part 11 Compliance
When it comes to 21CFR Part 11 compliance, if you are not compliant, you cannot use the data loggers and software, so it is imperative to know what regulations you need to meet and whether your software is compliant. 21CFR Part 11 compliance is great for pharmaceutical communities with regulatory compliance needs under the FDA that need to show they are meeting requirements on a regular basis. DT Pro software is 21CFR Part 11 compliant and has the ability to maintain regulatory compliance while processing and handling data.